|
Tris(ethylenediamine)cobalt(III) chloride in contrast to the bis(ethylenediamine) complexes does not undergo substitution. Give formulas corresponding to the following names: (a) Hexaaquachromium(llI) sulfate (b) Barium tetrabromoferrate(llI) (c) Bis(ethylenediamine)platinum(ll) carbonate chemistry Only a few lanthanides show an oxidation state other than +3. The trans isomer cation has idealized D 2h point group symmetry, whereas the cis isomer cation has C 2 symmetry. Both isomers of dichlorobis(ethylenediamine)cobalt(III) have often been used in stereochemical studies. The chiral cis isomer is obtained by heating the trans isomer. This pair of isomers were significant in the development of the area of coordination chemistry. This salt is more soluble than cis-dichlorobis(ethylenediamine)cobalt(III) chloride. Cl + 2 HCl → trans-Cl + CO 2 + H 2O UV-vis spectra of various stages in the conversion of trans- + to the cis isomer. Before Alfred Werner, in the 1890s, established the idea of a complex ion, coordination compounds had traditional formulas. Alternatively, (carbonato)bis(ethylenediamine)cobalt(III) chloride reacts with hydrochloric acid at 10 ☌ to give the same species. Chromium(III), like cobalt(III), has a coordination number of 6 in many of its complex ions. The initial product contains HCl, which is removed by heating. Coordination number: Ions: Dibromobis(ethylenediamine)cobalt(III) sulfate Hexaamminechromium(III) tetrachlorocuprate(II) 6 chromium in. Note that this (I presume) is a Co3+ complex, i.e. Write the number of individual ions per formula unit and the coordination number of the metal ion in each of the compounds below. Ethylenediamine is a bidentate ligand it has 2 donor atoms to coordinate to the metal centre in a 5-membered chelate ring. The compound is synthesized by the reaction of cobalt(II) chloride and ethylenediamine in hydrochloric acid in the presence of oxygen:Ĥ CoCl 2 + 8 en + 4 HCl + O 2 → 4 trans-Cl + 2 H 2O ignment 9 0 Help Saved 1 attempts left Check my work Be sure to answer all parts. 
The more stable trans-dichlorobis(ethylenediamine)cobalt(III) chloride is also known. One chloride ion in this salt readily undergoes ion exchange but the two other chlorides are less reactive, being bound to the metal center. It is the monochloride salt of the cationic coordination complex +. It is a green diamagnetic solid that is soluble in water.  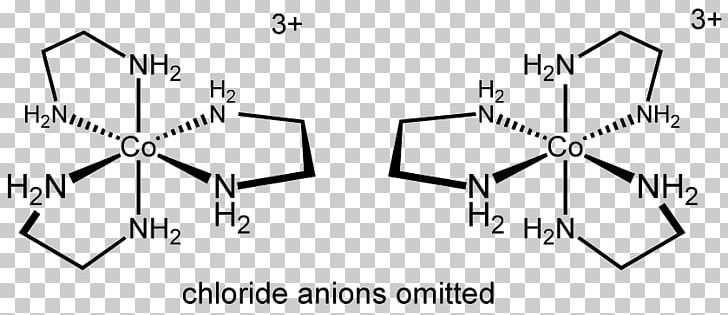
Trans-Dichlorobis(ethylenediamine)cobalt(III) chloride is a salt with the formula Cl (en = ethylenediamine).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
